-
 US, Iran trade threats to target infrastructure in Middle East
US, Iran trade threats to target infrastructure in Middle East
-
Paris doubles up with super-G victory at World Cup finals

-
 Dortmund part ways with sporting director Kehl
Dortmund part ways with sporting director Kehl
-
Russia resumes use of space launch site damaged in accident

-
 Cuba scrambles to restore power after new blackout
Cuba scrambles to restore power after new blackout
-
Senegal's Idrissa Gueye ready to 'hand back' AFCON medals

-
 New Zealand's Walsh bags fourth world indoor gold
New Zealand's Walsh bags fourth world indoor gold
-
Goggia claims first super-G title after victory in Kvitfjell

-
 Slovenia votes in tight polls, with conservatives eyeing comeback
Slovenia votes in tight polls, with conservatives eyeing comeback
-
A herd stop: Train kills 3 rare bison in Poland

-
 Vietnam, Russia to sign energy deal: Hanoi
Vietnam, Russia to sign energy deal: Hanoi
-
American Gumberg triumphs in Hainan for second DP World Tour win

-
 South Africa clinch 19-run win over New Zealand in fourth T20
South Africa clinch 19-run win over New Zealand in fourth T20
-
Iran threatens Middle East infrastructure after Trump ultimatum

-
 French elect mayors in key cities including Paris
French elect mayors in key cities including Paris
-
'They beat us with whips': Sudan RSF detainees tell of horrors in El-Fasher

-
 Australia's Hannah Green wins historic third tournament in a row
Australia's Hannah Green wins historic third tournament in a row
-
China's premier vows to expand global 'trade pie': state media

-
 Belgium commemorates Brussels attacks 10 years on
Belgium commemorates Brussels attacks 10 years on
-
Sri Lanka raises fuel prices by 25 percent as war bites

-
 Rights groups fear use of arrest to stifle free speech in Pakistan
Rights groups fear use of arrest to stifle free speech in Pakistan
-
Iranian missiles sow panic, destruction in Israeli towns

-
 Damaged Russian tanker to be towed to Libya: state-owned company
Damaged Russian tanker to be towed to Libya: state-owned company
-
Gilgeous-Alexander scores 40, LeBron breaks NBA appearance record

-
 Cuba hit by second nationwide blackout in a week
Cuba hit by second nationwide blackout in a week
-
BTS draws over 100,000 fans to Seoul comeback concert: label

-
 US-China 'Board of Trade' may help ties but experts flag market worries
US-China 'Board of Trade' may help ties but experts flag market worries
-
Sinner, defending champ Mensik advance to third round at Miami Open

-
 Iran missile strikes wound over 100 in two south Israel towns
Iran missile strikes wound over 100 in two south Israel towns
-
Shai hits 40 as Thunder win despite NBA melee with four ejected

-
 Records shattered as US heatwave moves eastward
Records shattered as US heatwave moves eastward
-
Iran missiles hit southern Israel, injuring more than 100

-
 LeBron James breaks record for most NBA games played
LeBron James breaks record for most NBA games played
-
'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Japan coach says Asian Cup crown 'well-deserved' for inspirational team
Japan coach says Asian Cup crown 'well-deserved' for inspirational team
-
PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
-
Milan move to within five points of Serie A leaders Inter

-
 Duplantis masterclass as Kerr and record-setter Ehammer shine
Duplantis masterclass as Kerr and record-setter Ehammer shine
-
Rosenior urges Chelsea to 'forget the noise' after damaging loss

-
 Marquez ambushed Di Giannantonio to win Brazil sprint
Marquez ambushed Di Giannantonio to win Brazil sprint
-
Sweden's Duplantis wins fourth world indoor pole vault title

-
 Liverpool, Chelsea slip up in Champions League race
Liverpool, Chelsea slip up in Champions League race
-
WHO sends first overland convoy from emergencies hub to Beirut

-
 Everton rub salt in Chelsea wounds as Champions League race tightens
Everton rub salt in Chelsea wounds as Champions League race tightens
-
Coach Mignoni returns but Toulon crash to Stade Francais

-
 Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
-
Sinner and Pegula advance to third round at Miami Open

-
 Britain's Kerr outsprints Hocker for world indoor 3,000m gold
Britain's Kerr outsprints Hocker for world indoor 3,000m gold
-
Kane backs Tuchel's call to rest him from England friendly


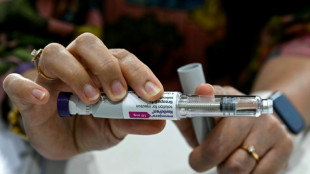
US approves new drug to treat Alzheimer's disease
The US Food and Drug Administration on Friday approved a highly anticipated new drug designed to slow cognitive decline in patients in mild and early stages of Alzheimer's disease.
The FDA approval of the drug, Leqembi, also known as lecanemab, comes just days after the regulatory agency was harshly criticized in a congressional report for its green-lighting of another Alzheimer's drug, Aduhelm.
And it was granted despite trial results showing the monoclonal antibody treatment carries risks of brain swelling and bleeding.
Both drugs were approved through an accelerated process that allows the FDA to fast-track approval of drugs for serious conditions where there is an unmet medical need.
Leqembi and Aduhelm, which were jointly developed by Japan's Eisai and Biogen of the United States, "represent an important advancement in the ongoing fight to effectively treat Alzheimer's disease," the FDA said in a statement.
"Alzheimer's disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones," Billy Dunn of the FDA's Center for Drug Evaluation and Research said in a statement.
Leqembi, Dunn said, is "the latest therapy to target and affect the underlying disease process of Alzheimer's, instead of only treating the symptoms of the disease."
Approximately 6.5 million Americans suffer from Alzheimer's, which is characterized by memory loss and declining mental acuity.
Preliminary data from a trial of Leqembi was released in September and found it slowed cognitive decline in Alzheimer's patients by 27 percent.
The phase three trial involved nearly 1,800 people, divided between those given the drug and given a placebo, and ran over 18 months.
The complete trial data, published in the New England Journal of Medicine, raised concern about the incidence of "adverse effects" including brain bleeds and swelling.
The results showed that 17.3 percent of patients administered the drug experienced brain bleeds, compared with nine percent of those receiving a placebo.
And 12.6 percent of those taking the drug experienced brain swelling, compared with just 1.7 percent of those in the placebo group.
Deaths were reported at approximately the same rate in both arms of the trial of the drug.
- $26,500 a year -
In Alzheimer's disease, two key proteins, tau and amyloid beta, build up into tangles and plaques, known together as aggregates, which cause brain cells to die and lead to brain shrinkage.
Leqembi, which is administered intravenously once every two weeks, works by targeting amyloid.
In the trial, patients receiving Leqembi had a statistically significant reduction in brain amyloid plaque compared to the placebo arm, which had no reduction of amyloid beta plaque.
Biogen and Eisai previously brought Aduhelm to market, but there was significant controversy over whether it worked, and its approval in 2021 led to three high-level resignations in the FDA.
A US congressional investigation said the accelerated approval process for Aduhelm, the first drug approved in decades to treat Alzheimer's, was "rife with irregularities" and criticized both the agency and Biogen.
The Cambridge, Massachusetts-based Biogen set an "unjustifiably high price" for Aduhelm of $56,000 a year, the congressional report said.
Eisai said Leqembi would be priced initially at $26,500 per year and estimated that 100,000 Americans could be receiving the drug by three years from now.
Joanne Pike, president and CEO of the Alzheimer's Association, welcomed the approval of Leqembi but expressed concern that its high cost could put it out of reach of most Americans, particularly if it is not covered by Medicare, the government health insurance program for the elderly.
"People living with this fatal disease today do not have time to wait for a miracle drug or cure," Pike said in a statement.
C.Amaral--PC