-
 French elect mayors in key cities including Paris
French elect mayors in key cities including Paris
-
'They beat us with whips': Sudan RSF detainees tell of horrors in El-Fasher

-
 Australia's Hannah Green wins historic third tournament in a row
Australia's Hannah Green wins historic third tournament in a row
-
China's premier vows to expand global 'trade pie': state media

-
 Belgium commemorates Brussels attacks 10 years on
Belgium commemorates Brussels attacks 10 years on
-
Sri Lanka raises fuel prices by 25 percent as war bites

-
 Rights groups fear use of arrest to stifle free speech in Pakistan
Rights groups fear use of arrest to stifle free speech in Pakistan
-
Iranian missiles sow panic, destruction in Israeli towns

-
 Damaged Russian tanker to be towed to Libya: state-owned company
Damaged Russian tanker to be towed to Libya: state-owned company
-
Gilgeous-Alexander scores 40, LeBron breaks NBA appearance record

-
 Cuba hit by second nationwide blackout in a week
Cuba hit by second nationwide blackout in a week
-
BTS draws over 100,000 fans to Seoul comeback concert: label

-
 US-China 'Board of Trade' may help ties but experts flag market worries
US-China 'Board of Trade' may help ties but experts flag market worries
-
Sinner, defending champ Mensik advance to third round at Miami Open

-
 Iran missile strikes wound over 100 in two south Israel towns
Iran missile strikes wound over 100 in two south Israel towns
-
Shai hits 40 as Thunder win despite NBA melee with four ejected

-
 Records shattered as US heatwave moves eastward
Records shattered as US heatwave moves eastward
-
Iran missiles hit southern Israel, injuring more than 100

-
 LeBron James breaks record for most NBA games played
LeBron James breaks record for most NBA games played
-
'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Japan coach says Asian Cup crown 'well-deserved' for inspirational team
Japan coach says Asian Cup crown 'well-deserved' for inspirational team
-
PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
-
Milan move to within five points of Serie A leaders Inter

-
 Duplantis masterclass as Kerr and record-setter Ehammer shine
Duplantis masterclass as Kerr and record-setter Ehammer shine
-
Rosenior urges Chelsea to 'forget the noise' after damaging loss

-
 Marquez ambushed Di Giannantonio to win Brazil sprint
Marquez ambushed Di Giannantonio to win Brazil sprint
-
Sweden's Duplantis wins fourth world indoor pole vault title

-
 Liverpool, Chelsea slip up in Champions League race
Liverpool, Chelsea slip up in Champions League race
-
WHO sends first overland convoy from emergencies hub to Beirut

-
 Everton rub salt in Chelsea wounds as Champions League race tightens
Everton rub salt in Chelsea wounds as Champions League race tightens
-
Coach Mignoni returns but Toulon crash to Stade Francais

-
 Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
-
Sinner and Pegula advance to third round at Miami Open

-
 Britain's Kerr outsprints Hocker for world indoor 3,000m gold
Britain's Kerr outsprints Hocker for world indoor 3,000m gold
-
Kane backs Tuchel's call to rest him from England friendly

-
 NBA fines 76ers' Drummond, Magic's Suggs $25,000 each
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each
-
Switzerland's Ehammer sets indoor heptathlon world record

-
 Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set
-
World Athletics decision to hand Asia two world indoors 'strategic' - Coe

-
 Trump threatens to use ICE agents for airport security control
Trump threatens to use ICE agents for airport security control
-
Kane moves closer to goals record as Bayern sink Union

-
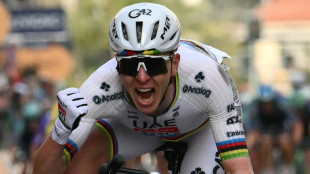 Pogacar ends long wait for Milan-San Remo glory after edging epic
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
US says 'took out' Iran base threatening blocked Hormuz oil route

-
 Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez
-
Welbeck scores twice to dent Liverpool's top-five hopes

-
 US strikes Iran bases threatening blocked Hormuz oil route
US strikes Iran bases threatening blocked Hormuz oil route
-
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin

-
 Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast
-
K-pop kings BTS stun Seoul in '2.0' comeback concert


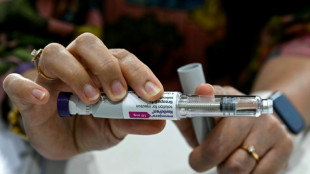
Biogen pulls controversial Alzheimer's drug Aduhelm
A controversial Alzheimer's drug that was trumpeted as the first to ever treat the cognitive decline associated with the devastating brain disorder has been pulled from the market, its maker Biogen announced Wednesday.
The US Food and Drug Administration awarded accelerated approval to Aduhelm in June 2021, a decision that was highly contentious at the time because the agency overruled its own independent advisors, who found there was insufficient evidence of benefit.
At least three of the 11-member independent committee that voted unanimously against recommending the drug subsequently resigned, and US congressional investigators slammed the accelerated approval as "rife with irregularities."
Biogen said it was discontinuing Aduhelm to put more resources into Leqembi, a newer Alzheimer's medicine that was fully approved last year under the traditional regulatory pathway.
"When searching for new medicines, one breakthrough can be the foundation that triggers future medicines to be developed," said Christopher Viehbacher, president and CEO of the Cambridge, Massachusetts-based biotech firm.
"Aduhelm was that groundbreaking discovery that paved the way for a new class of drugs and reinvigorated investments in the field."
Aduhelm, a monoclonal antibody that targets the build-up of a protein called amyloid beta in the brain tissue which is thought to be a cause of Alzheimer's, was tested in two late-stage human trials.
It showed a reduction in cognitive decline in one of the studies, but not the other.
According to a congressional report from December 2022, the FDA "considered Aduhelm under the traditional approval pathway used for most drugs for nine months, before abruptly changing course and granting approval under the accelerated approval pathway after a three-week review period."
The report said that FDA interactions with Biogen were "atypical" and included a failure to properly document contacts between agency staff and the drug maker.
The FDA and Biogen had also "inappropriately collaborated" on a joint briefing document for a key advisory committee, it said.
"FDA's approval process was rife with irregularities."
As for Biogen, the report said the company "viewed Aduhelm as an unprecedented financial opportunity -- estimating a potential peak revenue of $18 billion per year."
The congressional panel pointed to an "unjustifiably high price" for Aduhelm of $56,000 a year for patients.
Biogen's Leqembi, which it co-manufactures with Eisai of Japan, is now the only US approved treatment for Alzheimer's. It also targets amyloid beta and has been found to modestly reduce cognitive decline in patients with early stage disease.
Donanemab, developed by Eli Lilly, could be next to get the green light after performing similarly in clinical trials.
Alzheimer's is the most common form of dementia. More than one in nine people over 65 develop the condition, which worsens over time, robbing them of their memories and independence, according to the US Alzheimer's Association.
X.M.Francisco--PC