-
 Shai hits 40 as Thunder win despite NBA melee with four ejected
Shai hits 40 as Thunder win despite NBA melee with four ejected
-
Records shattered as US heatwave moves eastward

-
 Iran missiles hit southern Israel, injuring more than 100
Iran missiles hit southern Israel, injuring more than 100
-
LeBron James breaks record for most NBA games played

-
 'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1
'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1
-
Japan coach says Asian Cup crown 'well-deserved' for inspirational team

-
 PSG sweep past Nice to reclaim top spot in Ligue 1
PSG sweep past Nice to reclaim top spot in Ligue 1
-
Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81

-
 Milan move to within five points of Serie A leaders Inter
Milan move to within five points of Serie A leaders Inter
-
Duplantis masterclass as Kerr and record-setter Ehammer shine

-
 Rosenior urges Chelsea to 'forget the noise' after damaging loss
Rosenior urges Chelsea to 'forget the noise' after damaging loss
-
Marquez ambushed Di Giannantonio to win Brazil sprint

-
 Sweden's Duplantis wins fourth world indoor pole vault title
Sweden's Duplantis wins fourth world indoor pole vault title
-
Liverpool, Chelsea slip up in Champions League race

-
 WHO sends first overland convoy from emergencies hub to Beirut
WHO sends first overland convoy from emergencies hub to Beirut
-
Everton rub salt in Chelsea wounds as Champions League race tightens

-
 Coach Mignoni returns but Toulon crash to Stade Francais
Coach Mignoni returns but Toulon crash to Stade Francais
-
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81

-
 Sinner and Pegula advance to third round at Miami Open
Sinner and Pegula advance to third round at Miami Open
-
Britain's Kerr outsprints Hocker for world indoor 3,000m gold

-
 Kane backs Tuchel's call to rest him from England friendly
Kane backs Tuchel's call to rest him from England friendly
-
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each

-
 Switzerland's Ehammer sets indoor heptathlon world record
Switzerland's Ehammer sets indoor heptathlon world record
-
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set

-
 World Athletics decision to hand Asia two world indoors 'strategic' - Coe
World Athletics decision to hand Asia two world indoors 'strategic' - Coe
-
Trump threatens to use ICE agents for airport security control

-
 Kane moves closer to goals record as Bayern sink Union
Kane moves closer to goals record as Bayern sink Union
-
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
 US says 'took out' Iran base threatening blocked Hormuz oil route
US says 'took out' Iran base threatening blocked Hormuz oil route
-
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez

-
 Welbeck scores twice to dent Liverpool's top-five hopes
Welbeck scores twice to dent Liverpool's top-five hopes
-
US strikes Iran bases threatening blocked Hormuz oil route

-
 Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
-
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast

-
 K-pop kings BTS stun Seoul in '2.0' comeback concert
K-pop kings BTS stun Seoul in '2.0' comeback concert
-
French prosecutors suspect Musk encouraged deepfakes row to inflate X value

-
 Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
-
Iranians mark Eid as Tehran reports strike on nuclear plant

-
 Kenya, Uganda open rail extension burdened by Chinese debt
Kenya, Uganda open rail extension burdened by Chinese debt
-
K-pop kings BTS rock Seoul in comeback concert

-
 Invincible Japan edge Australia to win Women's Asian Cup
Invincible Japan edge Australia to win Women's Asian Cup
-
Italy's Paris claims first win of season in World Cup downhill finale

-
 In Finland, divers learn to explore icy polar waters
In Finland, divers learn to explore icy polar waters
-
Dortmund extend injured captain Can's contract

-
 Iranians mark Eid as Trump mulls winding down war
Iranians mark Eid as Trump mulls winding down war
-
Matisse's last years cut out -- but not pasted -- at Paris expo

-
 BTS fans take over central Seoul for K-pop kings' comeback
BTS fans take over central Seoul for K-pop kings' comeback
-
Star jockey McDonald becomes horse racing's most prolific Group 1 winner

-
 Israel strikes Tehran, Beirut as Trump mulls 'winding down' war
Israel strikes Tehran, Beirut as Trump mulls 'winding down' war
-
Pistons top Warriors to clinch NBA playoff berth

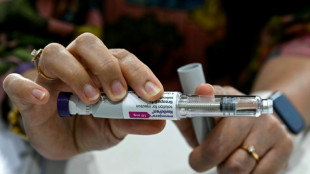
US health experts vote against MDMA as treatment for PTSD
A panel of US health experts convened by the Food and Drug Administration on Tuesday overwhelmingly voted against the use of MDMA, commonly known as ecstasy, to treat post-traumatic stress disorder.
PTSD, a debilitating mental health condition that develops after a person experiences or is threatened by traumatic events such as death, combat or sexual assault, affects an estimated five percent of Americans in any given year.
But pharmaceutical treatment options are so far limited to two antidepressants that require three months of dosing to take effect, and response rates to the medications have been found to be uneven.
California-based Lykos Therapeutics has based its request for regulatory approval on two clinical studies, each of which enrolled around 100 people, to evaluate MDMA used together with other psychological interventions such as talk therapy, against a placebo with talk therapy.
These two studies, published in the prestigious journal Nature Medicine, indicated MDMA was indeed both safe and highly effective at treating PTSD.
But nine out of 11 experts on the panel said available data was not enough to show the treatment was effective, and 10 out of 11 said the benefits did not outweigh the risks.
"I think this is a really exciting treatment. I'm really encouraged by the results to date," said one of the experts, Paul Holtzheimer of the National Center for PTSD.
"But I feel that both from an efficacy and a safety standpoint, it is still premature."
The vote by the panel of experts is non-binding, but the FDA rarely goes against their recommendations.
- Not enough side effect data -
MDMA -- methylenedioxymethamphetamine -- is a Schedule 1 drug under the Controlled Substances Act, and approving it for medical use would have represented a major shift.
In a briefing document put together ahead of the meeting, FDA staff raised concerns that although the studies were nominally "double-blinded" -- meaning neither those being tested nor their health care workers knew who received the treatment versus the placebo -- most people were able to accurately guess what they received.
This "functional unblinding," they argued, introduces bias and uncertainty into study outcomes.
FDA staff also criticized Lykos for not gathering sufficient side effect data, including whether participants experienced "euphoria" or "elated mood," which, they argued, "would be informative for an assessment of abuse potential or characterization of anticipated effects of the drug."
Reports from recreational use suggest MDMA has harmful impacts on heart and liver health -- but the company did not gather enough data in these areas, the FDA said.
- Research misconduct allegations -
Then there were troubling allegations of research impropriety linked to the trials that surfaced in a draft report by the nonprofit Institute for Clinical and Economic Review.
These include claims that the trials "pulled heavily from the existing community of those interested and involved in the use of psychedelics for possible psychological benefits."
Some patients told the nonprofit they were allegedly prevented from entering the long term-follow up study "and felt this was done to keep these negative outcomes out of the data set."
"We certainly take those allegations very seriously and are quite concerned by them," the FDA's Tiffany Farchione said during the meeting, adding a probe was ongoing.
Lykos has said a final decision from the FDA on authorization should come by mid-August.
If the agency were to authorize the treatment over the advisory panel's objections, there could be certain strict stipulations attached -- for example, that the drug only be dispensed in health care settings, with subjects carefully monitored and enrolled in a registry to document side effects.
X.Matos--PC