
-
 Former Russian insider says fear pushed elites to embrace Putin war
Former Russian insider says fear pushed elites to embrace Putin war
-
Evacuations 'ongoing' from hantavirus-hit cruise ship

-
 Oil tumbles and stocks rally on peace hopes, Samsung tops $1 trillion
Oil tumbles and stocks rally on peace hopes, Samsung tops $1 trillion
-
Asia football fans sweat on broadcast rights as World Cup nears

-
 US pauses Hormuz escorts, Trump says progress on Iran deal
US pauses Hormuz escorts, Trump says progress on Iran deal
-
Cambodian PM's cousin says owned 30% of scam-linked firm

-
 Hegseth's church brings its Christian nationalism to Washington
Hegseth's church brings its Christian nationalism to Washington
-
Afrobeats' Tiwa Savage nurtures Africa's future talent

-
 Venice Biennale opens in turmoil over Russian presence
Venice Biennale opens in turmoil over Russian presence
-
Philips profits double in first quarter

-
 Strasbourg on verge of European final amid fan displeasure at owners BlueCo
Strasbourg on verge of European final amid fan displeasure at owners BlueCo
-
Tradition, Trump and tennis: Five things about Pope Leo

-
 100 years on Earth: Iconic naturalist Attenborough marks century
100 years on Earth: Iconic naturalist Attenborough marks century
-
Bondi Beach mass shooting accused faces 19 extra charges

-
 Ukraine reports strike as Kyiv's ceasefire due to begin
Ukraine reports strike as Kyiv's ceasefire due to begin
-
Australia says 13 citizens linked to alleged IS members returning from Syria

-
 Thunder overpower Lakers, Pistons down Cavs
Thunder overpower Lakers, Pistons down Cavs
-
Boycott-hit 70th Eurovision celebrated under high security

-
 Court case challenges New Zealand's 'magical thinking' climate plans
Court case challenges New Zealand's 'magical thinking' climate plans
-
Iran war jolts China's well-oiled manufacturing hub

-
 Oil sinks and stocks rally on peace hopes, Samsung tops $1 trillion
Oil sinks and stocks rally on peace hopes, Samsung tops $1 trillion
-
Infantino defends World Cup ticket prices

-
 Pistons hold off Cavs to win series-opener
Pistons hold off Cavs to win series-opener
-
Rubio rising? Duel with Vance for 2028 heats up

-
 Teen shooter kills two at Brazil school
Teen shooter kills two at Brazil school
-
US pauses Hormuz escorts in bid for deal, as threats continue

-
 Judge orders German car-ramming suspect to psychiatric hospital
Judge orders German car-ramming suspect to psychiatric hospital
-
Fresh UAE attacks blamed on Iran draw new reality in the Gulf

-
 Case IQ Advances Its Leadership Position in AI For Investigations with Playbooks
Case IQ Advances Its Leadership Position in AI For Investigations with Playbooks
-
Transoft Solutions Acquires CADaptor Solutions

-
 Arsenal on cusp of history after reaching Champions League final
Arsenal on cusp of history after reaching Champions League final
-
Trump says pausing Hormuz operation in push for Iran deal

-
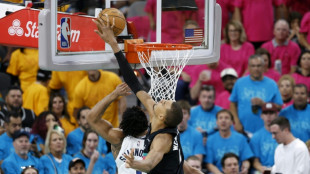 Wembanyama accused of 'obvious' illegal blocking
Wembanyama accused of 'obvious' illegal blocking
-
Musk 'was going to hit me,' OpenAI executive says at trial

-
 NFL star Diggs cleared of assaulting personal chef
NFL star Diggs cleared of assaulting personal chef
-
Fans 'set the standards' at rocking Emirates: Arteta

-
 Rubio warns against 'destabilizing' acts on Taiwan before Trump China visit
Rubio warns against 'destabilizing' acts on Taiwan before Trump China visit
-
US declares Iran offensive over, warns force remains an option

-
 Saka ends Arsenal's 20-year wait to reach Champions League final
Saka ends Arsenal's 20-year wait to reach Champions League final
-
Outgoing Costa Rica leader secures top post in new cabinet

-
 Rubio plays down Trump attacks on pope before Vatican trip
Rubio plays down Trump attacks on pope before Vatican trip
-
LIV Golf boss sees hope for new sponsors beyond 2026

-
 Mexican BTS fans go wild as concerts grow near
Mexican BTS fans go wild as concerts grow near
-
Europe's first commercial robotaxi service rolls out in Croatia

-
 Russian strikes kill 21 in Ukraine
Russian strikes kill 21 in Ukraine
-
Suspected hantavirus cases to be evacuated from cruise ship

-
 G7 trade ministers meet, not expected to discuss US tariff threat
G7 trade ministers meet, not expected to discuss US tariff threat
-
Hollywood star Malkovich gets Croatian citizenship

-
 Mickelson pulls out of PGA Championship for family issues
Mickelson pulls out of PGA Championship for family issues
-
Wales rugby great Halfpenny to retire


US health regulator rejects MDMA treatment for PTSD, for now
US health regulators on Friday denied an application for treating post-traumatic stress disorder (PTSD) with the drug MDMA, commonly known as ecstasy, saying more investigation needed to be done.
The company that submitted the application, Lykos Therapeutics, said in a statement that the Food and Drug Administration (FDA) had requested an additional Phase 3 clinical trial to study MDMA's "safety and efficacy."
A panel of experts convened by the FDA to evaluate clinical data on MDMA had overwhelmingly voted in early June to say there was insufficient evidence to prove it was effective.
While unsurprising, the decision announced Friday represents a blow to advocates of the novel treatment.
"The FDA request for another study is deeply disappointing, not just for all those who dedicated their lives to this pioneering effort, but principally for the millions of Americans with PTSD... who have not seen any new treatment options in over two decades, said Lykos CEO Amy Emerson.
PTSD is a debilitating mental health condition that develops after a person experiences or is threatened by traumatic events such as death, combat or sexual assault.
It affects an estimated five percent of Americans in any given year.
Pharmaceutical treatment options for PTSD are so far limited to two antidepressants that require three months of dosing to take effect, and response rates to the medications have been found to be uneven.
MDMA -- methylenedioxymethamphetamine -- is a Schedule 1 drug under the Controlled Substances Act, and approving it for medical use would have represented a major shift.
California-based Lykos based its request for regulatory approval on two clinical studies, each of which enrolled around 100 people, to evaluate MDMA used together with other psychological interventions such as talk therapy, against a placebo with talk therapy.
These two studies, published in the prestigious journal Nature Medicine, indicated MDMA was indeed both safe and highly effective at treating PTSD.
But nine out of 11 experts on the FDA panel said available data was not enough to show the treatment was effective, and 10 out of 11 said the benefits did not outweigh the risks.
In a briefing document put together ahead of the meeting, FDA staff raised concerns about Lykos's clinical trial methodology and criticized the company for not gathering sufficient side effect data.
The company said it will "work diligently in the coming months to address FDA's concerns and to take advantage of agency processes to resolve scientific disagreements."
"We intend to work tirelessly and use all available regulatory pathways to find a reasonable and expeditious path forward," Emerson added.
M.Carneiro--PC




