-
 Strasbourg on verge of European final amid fan displeasure at owners BlueCo
Strasbourg on verge of European final amid fan displeasure at owners BlueCo
-
Tradition, Trump and tennis: Five things about Pope Leo

-
 100 years on Earth: Iconic naturalist Attenborough marks century
100 years on Earth: Iconic naturalist Attenborough marks century
-
Bondi Beach mass shooting accused faces 19 extra charges

-
 Ukraine reports strike as Kyiv's ceasefire due to begin
Ukraine reports strike as Kyiv's ceasefire due to begin
-
Australia says 13 citizens linked to alleged IS members returning from Syria

-
 Thunder overpower Lakers, Pistons down Cavs
Thunder overpower Lakers, Pistons down Cavs
-
Boycott-hit 70th Eurovision celebrated under high security

-
 Court case challenges New Zealand's 'magical thinking' climate plans
Court case challenges New Zealand's 'magical thinking' climate plans
-
Iran war jolts China's well-oiled manufacturing hub

-
 Oil sinks and stocks rally on peace hopes, Samsung tops $1 trillion
Oil sinks and stocks rally on peace hopes, Samsung tops $1 trillion
-
Infantino defends World Cup ticket prices

-
 Pistons hold off Cavs to win series-opener
Pistons hold off Cavs to win series-opener
-
Rubio rising? Duel with Vance for 2028 heats up

-
 Teen shooter kills two at Brazil school
Teen shooter kills two at Brazil school
-
US pauses Hormuz escorts in bid for deal, as threats continue

-
 Judge orders German car-ramming suspect to psychiatric hospital
Judge orders German car-ramming suspect to psychiatric hospital
-
Fresh UAE attacks blamed on Iran draw new reality in the Gulf

-
 Arsenal on cusp of history after reaching Champions League final
Arsenal on cusp of history after reaching Champions League final
-
Trump says pausing Hormuz operation in push for Iran deal

-
 Wembanyama accused of 'obvious' illegal blocking
Wembanyama accused of 'obvious' illegal blocking
-
Musk 'was going to hit me,' OpenAI executive says at trial

-
 NFL star Diggs cleared of assaulting personal chef
NFL star Diggs cleared of assaulting personal chef
-
Fans 'set the standards' at rocking Emirates: Arteta

-
 Rubio warns against 'destabilizing' acts on Taiwan before Trump China visit
Rubio warns against 'destabilizing' acts on Taiwan before Trump China visit
-
US declares Iran offensive over, warns force remains an option

-
 Saka ends Arsenal's 20-year wait to reach Champions League final
Saka ends Arsenal's 20-year wait to reach Champions League final
-
Outgoing Costa Rica leader secures top post in new cabinet

-
 Rubio plays down Trump attacks on pope before Vatican trip
Rubio plays down Trump attacks on pope before Vatican trip
-
LIV Golf boss sees hope for new sponsors beyond 2026

-
 Mexican BTS fans go wild as concerts grow near
Mexican BTS fans go wild as concerts grow near
-
Europe's first commercial robotaxi service rolls out in Croatia

-
 Russian strikes kill 21 in Ukraine
Russian strikes kill 21 in Ukraine
-
Suspected hantavirus cases to be evacuated from cruise ship

-
 G7 trade ministers meet, not expected to discuss US tariff threat
G7 trade ministers meet, not expected to discuss US tariff threat
-
Hollywood star Malkovich gets Croatian citizenship

-
 Mickelson pulls out of PGA Championship for family issues
Mickelson pulls out of PGA Championship for family issues
-
Wales rugby great Halfpenny to retire

-
 Rahm says player concessions needed to save LIV Golf
Rahm says player concessions needed to save LIV Golf
-
Bowlers, Samson keep Chennai afloat in IPL playoff race

-
 Rolling Stones announce July 10 release of new album 'Foreign Tongues'
Rolling Stones announce July 10 release of new album 'Foreign Tongues'
-
France's Macron taps ex-aide to head central bank

-
 PSG 'not here to defend' against Bayern, says Luis Enrique
PSG 'not here to defend' against Bayern, says Luis Enrique
-
Trump says he works out 'one minute a day' as he restores fitness award

-
 Russia hits Ukraine with deadly strikes as Zelensky denounces Moscow's 'cynicism'
Russia hits Ukraine with deadly strikes as Zelensky denounces Moscow's 'cynicism'
-
EU urges US to stick to tariff deal terms

-
 Hantavirus on the Hondius: what we know
Hantavirus on the Hondius: what we know
-
Rahm eligible for Ryder Cup after deal with European Tour

-
 Stocks rise, oil falls as traders eye earnings, US-Iran ceasefire
Stocks rise, oil falls as traders eye earnings, US-Iran ceasefire
-
Bayern's Kompany channels 'inner tranquility' before PSG showdown


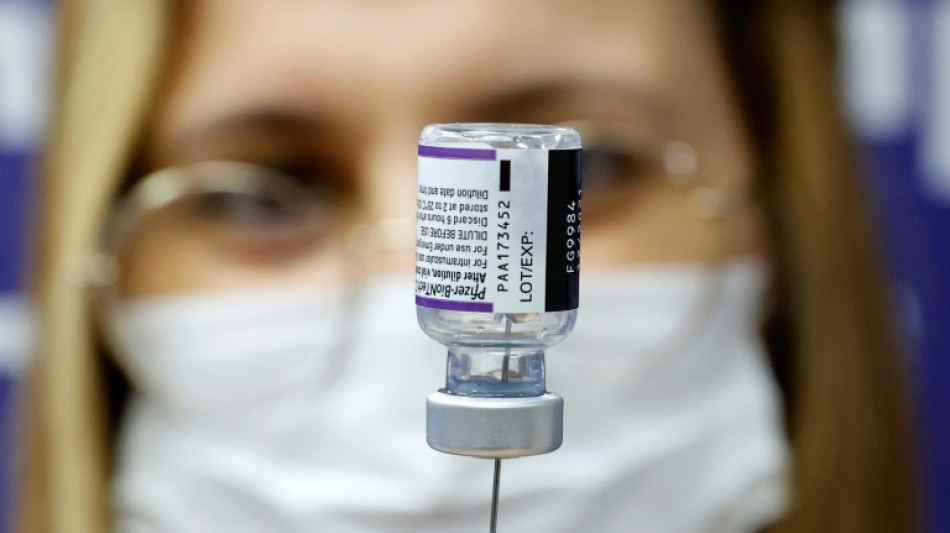
Pfizer Covid vaccine for under-fives effective with three doses
The Pfizer/BioNTech Covid vaccine is safe and effective for children aged six months to under five years when given in three doses, the companies said in a statement Monday.
The announcement comes as the US Food and Drug Administration (FDA) is planning meetings in the coming weeks to weigh authorizing Covid vaccines among the youngest children, the only age group who are not yet eligible in most countries, a source of concern to many parents.
Pfizer/BioNTech evaluated three doses, given at three micrograms, in a clinical trial and found the vaccine evoked a strong immune response. Side effects were similar in the vaccine and placebo groups.
Vaccine efficacy was 80.3 percent, according to a preliminary estimate.
"We are pleased that our formulation for the youngest children, which we carefully selected to be one-tenth of the dose strength for adults, was well tolerated and produced a strong immune response," said Pfizer CEO Albert Bourla in a statement.
"We look forward to soon completing our submissions to regulators globally with the hope of making this vaccine available to younger children as quickly as possible, subject to regulatory authorization," he added.
The FDA has tentatively scheduled three dates in June where experts will meet and likely decide whether to authorize the Pfizer Covid vaccine for under-fives and the Moderna vaccine for under-sixes, which is given as two shots of 25 micrograms.
The agency was originally set to evaluate the Pfizer vaccine given as two doses in February, but data showed it did not provoke a strong enough immune response in children aged two to four. The FDA then asked to see data for a third shot.
- Data welcomed -
According to the new data, 1,678 children received a third dose at least two months after the second dose, at a time when Omicron was the predominant variant.
An analysis of a subset of participants showed antibody levels were similar to 16- to 25-year-olds who were given the full strength vaccine at two doses.
No new adverse events were identified, and the majority of side effects were mild or moderate.
"Three doses of (Pfizer's) Covid vaccine appear to be very safe and highly effective in preventing not only severe disease, hospitalization, and death from Covid, but even symptomatic Covid at a time when Omicron was the dominant variant," Celine Gounder, editor-at-large for public health at Kaiser Health News told AFP.
"However, we know that protection against SARS-CoV-2 infection and milder symptomatic disease wanes over time," added Gounder, an infectious disease specialist and epidemiologist.
"Pfizer is reporting follow-up data only out to seven days after the third dose of vaccine. It's too early to say how the three-dose series would perform out to several months or a year."
Jeremy Faust, of Brigham and Women's Hospital Department of Emergency Medicine, told AFP: "My first impression is very positive. These numbers are exactly the kinds of signals we wanted to see."
"I wish the two-dose series had worked for Pfizer/BioNTech. It didn't. But the three-dose series appears to have given these very young children the protection we want them to have," the doctor added.
If and when both Pfizer's and Moderna's vaccines are authorized, US parents will have to consider whether they want their children to receive Moderna's two dose vaccine -- which will offer faster protection -- or Pfizer's three doses -- which will take longer to be effective but may ultimately be more protective.
Pfizer's announcement comes one week after US health authorities gave the green light for the company's Covid booster shots to be administered to children age five to 11.
Severe disease from Covid is very rare among under-fives but can occur, with 477 US deaths in this age group since the start of the pandemic, or about 0.1 percent of all deaths.
Children can also contract a rare post-viral condition called multisystem inflammatory syndrome in children (MIS-C), which has affected some 8,210 US children and killed 68.
Like adults, some children who get Covid may go on to develop long Covid, with new, ongoing or returning symptoms, including brain fog and fatigue.
A.S.Diogo--PC


