
-
 Arsenal on cusp of history after reaching Champions League final
Arsenal on cusp of history after reaching Champions League final
-
Trump says pausing Hormuz operation in push for Iran deal

-
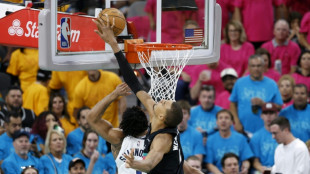 Wembanyama accused of 'obvious' illegal blocking
Wembanyama accused of 'obvious' illegal blocking
-
Musk 'was going to hit me,' OpenAI executive says at trial

-
 NFL star Diggs cleared of assaulting personal chef
NFL star Diggs cleared of assaulting personal chef
-
Fans 'set the standards' at rocking Emirates: Arteta

-
 Rubio warns against 'destabilizing' acts on Taiwan before Trump China visit
Rubio warns against 'destabilizing' acts on Taiwan before Trump China visit
-
US declares Iran offensive over, warns force remains an option

-
 Saka ends Arsenal's 20-year wait to reach Champions League final
Saka ends Arsenal's 20-year wait to reach Champions League final
-
Outgoing Costa Rica leader secures top post in new cabinet

-
 Rubio plays down Trump attacks on pope before Vatican trip
Rubio plays down Trump attacks on pope before Vatican trip
-
LIV Golf boss sees hope for new sponsors beyond 2026

-
 Mexican BTS fans go wild as concerts grow near
Mexican BTS fans go wild as concerts grow near
-
Europe's first commercial robotaxi service rolls out in Croatia

-
 Russian strikes kill 21 in Ukraine
Russian strikes kill 21 in Ukraine
-
Suspected hantavirus cases to be evacuated from cruise ship

-
 G7 trade ministers meet, not expected to discuss US tariff threat
G7 trade ministers meet, not expected to discuss US tariff threat
-
Hollywood star Malkovich gets Croatian citizenship

-
 Mickelson pulls out of PGA Championship for family issues
Mickelson pulls out of PGA Championship for family issues
-
Wales rugby great Halfpenny to retire

-
 Rahm says player concessions needed to save LIV Golf
Rahm says player concessions needed to save LIV Golf
-
Bowlers, Samson keep Chennai afloat in IPL playoff race

-
 Rolling Stones announce July 10 release of new album 'Foreign Tongues'
Rolling Stones announce July 10 release of new album 'Foreign Tongues'
-
France's Macron taps ex-aide to head central bank

-
 PSG 'not here to defend' against Bayern, says Luis Enrique
PSG 'not here to defend' against Bayern, says Luis Enrique
-
Trump says he works out 'one minute a day' as he restores fitness award

-
 Russia hits Ukraine with deadly strikes as Zelensky denounces Moscow's 'cynicism'
Russia hits Ukraine with deadly strikes as Zelensky denounces Moscow's 'cynicism'
-
EU urges US to stick to tariff deal terms

-
 Hantavirus on the Hondius: what we know
Hantavirus on the Hondius: what we know
-
Rahm eligible for Ryder Cup after deal with European Tour

-
 Stocks rise, oil falls as traders eye earnings, US-Iran ceasefire
Stocks rise, oil falls as traders eye earnings, US-Iran ceasefire
-
Bayern's Kompany channels 'inner tranquility' before PSG showdown

-
 Colombian mine explosion kills nine
Colombian mine explosion kills nine
-
Matthews latest England World Cup-winner out of Women's Six Nations

-
 Race to find port for cruise ship battling deadly rodent virus
Race to find port for cruise ship battling deadly rodent virus
-
Celtic's O'Neill says Hearts' rise good for Scottish football

-
 Ethiopia and Sudan accuse each other of attacks
Ethiopia and Sudan accuse each other of attacks
-
Injured Mbappe faces backlash over Sardinia trip before Clasico

-
 Vodafone to take full ownership of UK mobile operator
Vodafone to take full ownership of UK mobile operator
-
Stocks advance, oil falls as traders eye US-Iran ceasefire

-
 Sabalenka ready to boycott Grand Slams over prize money
Sabalenka ready to boycott Grand Slams over prize money
-
Boko Haram attack on Chad army base kills at least 24: military, local officials

-
 US trade gap widens in March as AI spending boosts imports
US trade gap widens in March as AI spending boosts imports
-
US threatens 'devastating' response to any Iran attack on shipping

-
 Murphy warns snooker hopefuls to 'work harder' to match Chinese stars
Murphy warns snooker hopefuls to 'work harder' to match Chinese stars
-
Race to find port for hantavirus-stricken cruise ship

-
 Romanian pro-EU PM loses no-confidence motion
Romanian pro-EU PM loses no-confidence motion
-
Edin Terzic to become Athletic Bilbao coach next season

-
 Borthwick backed by RFU to take England to 2027 Rugby World Cup
Borthwick backed by RFU to take England to 2027 Rugby World Cup
-
EU hails 'leap forward' in ties with Russia's ally Armenia


Pfizer to seek US authorization for third Covid shot in children
Pfizer and BioNTech on Thursday announced positive results from a clinical trial on the safety and immune response of a third dose of their Covid vaccine in children aged five through 11, adding they would soon seek regulatory authorization.
Third doses of the vaccine are recommended for those aged 12 and up, and a fourth dose was recently recommended for people over 50.
Younger children -- except for those with immune compromising conditions -- have not been eligible for the third, making them more susceptible to infection from Omicron and its BA.2 subvariant.
BA.2 is now the globally dominant strain, and is behind a current spike in cases in the northeastern United States.
In the phase 2/3 trial, the companies analyzed data from 140 children aged five through 11, approximately six months after the second dose.
The dosage in this group is 10 micrograms, which was selected for safety reasons as children are more susceptible to side effects. The dose for those 12 and up is 30 micrograms.
Across the 140 children analyzed, the third dose was well tolerated, revealing no new safety concerns.
They also analyzed blood sera from a group of 30 individuals, finding that a third dose caused a 36-fold increase in levels of infection-blocking neutralizing antibodies against Omicron, compared to two doses.
Pfizer and BioNTech plan to soon submit the data to the US Food and Drug Administration (FDA), the European Medicines Agency and other regulatory agencies.
Most countries, including the United States, haven't yet authorized Covid vaccines for infants and very young children.
Last month, Moderna said it was pursuing approval for its vaccine in children aged six months through five years, using a two-dose regimen.
Pfizer's vaccine for this group was meant to be considered by the FDA in February but the agency postponed the meeting, because it wanted more data on how it would perform with three doses.
A.Magalhes--PC

