-
 Palace overcome Shakhtar to reach historic Conference League final
Palace overcome Shakhtar to reach historic Conference League final
-
Watkins salutes Emery after Villa reach Europa final

-
 AI actors not eligible for Golden Globes, say organizers
AI actors not eligible for Golden Globes, say organizers
-
Kuebler brace sends Freiburg past Braga into Europa League final

-
 Rayo down Strasbourg in Conference League to set up first European final
Rayo down Strasbourg in Conference League to set up first European final
-
Villa crush Forest to reach Europa League final against Freiburg

-
 Brazil's Lula and Trump hail positive talks after rocky relations
Brazil's Lula and Trump hail positive talks after rocky relations
-
Shakira teases new World Cup song

-
 Palace beat Shakhtar to reach first European final
Palace beat Shakhtar to reach first European final
-
Rail fare to World Cup final stadium is cut ... to $105

-
 Global stocks mostly fall as US rally shows signs of fatigue
Global stocks mostly fall as US rally shows signs of fatigue
-
Sabalenka, champion Paolini open Italian Open accounts

-
 Trump gives EU until July 4 to ratify deal or face tariff hike
Trump gives EU until July 4 to ratify deal or face tariff hike
-
30 passengers left hantavirus ship in Saint Helena: cruise operator

-
 Real Madrid to punish Valverde, Tchouameni after training ground clash
Real Madrid to punish Valverde, Tchouameni after training ground clash
-
French parliament votes to ease returns of looted art to ex-colonies

-
 Ancelotti set for Brazil contract extension: federation
Ancelotti set for Brazil contract extension: federation
-
Civilians lynched in Mali witch hunt after jihadist, rebel attacks

-
 US targets Cuban military, mine in new sanctions
US targets Cuban military, mine in new sanctions
-
Marsh ton sets up Lucknow win in rain-hit IPL clash

-
 Google faces new UK lawsuit over online display ads
Google faces new UK lawsuit over online display ads
-
Yankees outfielder Dominguez collides with wall making catch

-
 NY to hire 500 addiction recovery mentors with opioid settlement cash
NY to hire 500 addiction recovery mentors with opioid settlement cash
-
Trump says he would not pay $1,000 to watch US at World Cup

-
 Dubois vows to take out 'trash' WBO heavyweight champion Wardley
Dubois vows to take out 'trash' WBO heavyweight champion Wardley
-
France to ban CBD edibles: sources

-
 Twin jihadist-claimed attacks kill more than 30 in Mali
Twin jihadist-claimed attacks kill more than 30 in Mali
-
US oil blockade on Cuba 'energy starvation': UN experts
-
 Zelensky warns against attending Russia's parade as Moscow repeats threats
Zelensky warns against attending Russia's parade as Moscow repeats threats
-
Millwall eye 'fairytale' in Championship play-offs

-
 Hantavirus not like Covid: doctor treating patient in Netherlands
Hantavirus not like Covid: doctor treating patient in Netherlands
-
Covid flashbacks haunt Canary Islands as hantavirus ship nears

-
 IOC lifts Olympic ban on Belarus but Russia 'still suspended'
IOC lifts Olympic ban on Belarus but Russia 'still suspended'
-
IMF warns of 'inevitable' AI-powered threats to global financial system

-
 Brighton boss Hurzeler agrees new three-year deal
Brighton boss Hurzeler agrees new three-year deal
-
WHO says now five confirmed cruise ship hantavirus cases

-
 Spurs boss De Zerbi shrugs off criticism of win over weakened Villa
Spurs boss De Zerbi shrugs off criticism of win over weakened Villa
-
Sinner demands 'respect' from Grand Slams, Djokovic lends support in prize money row

-
 Germany warns tax revenues to be hit by Iran war
Germany warns tax revenues to be hit by Iran war
-
Italy's tennis chief wants to break Grand Slam 'monopoly' with new major

-
 IOC rules out 'crossover' sports at 2030 Winter Olympics
IOC rules out 'crossover' sports at 2030 Winter Olympics
-
WHO warns of more hantavirus cases in 'limited' outbreak

-
 Real Madrid's Valverde treated in hospital after Tchouameni clash: reports
Real Madrid's Valverde treated in hospital after Tchouameni clash: reports
-
Past hantavirus outbreak shows how Andes virus spreads

-
 EU prosecutors probe alleged misuse of funds linked to France's Bardella
EU prosecutors probe alleged misuse of funds linked to France's Bardella
-
UK police officers probed over handling of Al-Fayed complaints

-
 Paolini begins Italian Open title defence by battling past Jeanjean
Paolini begins Italian Open title defence by battling past Jeanjean
-
Brazil must channel World Cup pressure into motivation: Luiz Henrique

-
 AI use surges globally but rich-poor divide widens, Microsoft says
AI use surges globally but rich-poor divide widens, Microsoft says
-
Carrick says strong finish matters more than his Man Utd future


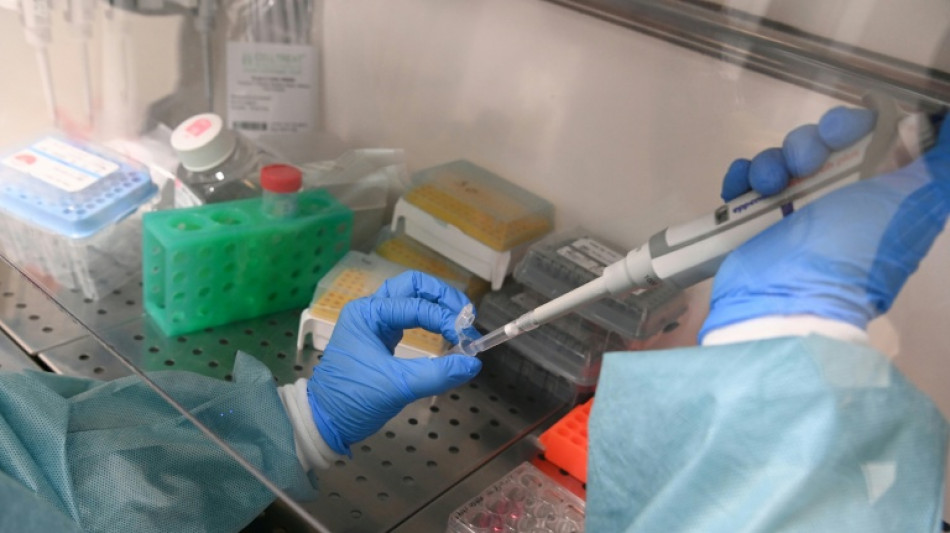
In reversal, US agrees to review new Moderna flu shot
Vaccine manufacturer Moderna said Wednesday the US Food and Drug Administration walked back its previous position and agreed to review the company's new mRNA-based flu shot.
Last week the US firm said the federal vaccine regulator rejected the application for review of the new shot, calling its clinical trial inadequate.
But Moderna said that after a "constructive" meeting the FDA had accepted the application for review based on a regulatory pathway focused on older adults.
The company's application now seeks full approval for adults 50 to 64 and accelerated approval for adults 65 and older, Moderna said in a statement. It also agreed to conduct an additional study after the shot hits the market.
The FDA's rejection had come as the body has called for a reconsideration of approval procedures for certain vaccines, including for influenza -- proposed federal policy changes under President Donald Trump that have triggered widespread alarm among public health and medical professionals.
The new shot uses mRNA technology, which health chief Robert F. Kennedy Jr, a vocal vaccine skeptic, has criticized. He notably cut off federal research grants that funded mRNA development.
That contradicts Trump's position during his first presidential term, when he called mRNA technology a "modern-day miracle."
It was used during the Covid-19 pandemic to swiftly develop an immunization that global health authorities deemed safe and effective against the fast-spreading illness. It was credited with saving millions of lives.
Moderna's new shot had already been accepted for review in the European Union, Canada and Australia.
Vinay Prasad, the top US vaccine official, had signed the letter rejecting Moderna's bid for approval, saying the company's clinical trial was not "adequate and well-controlled," and had not tested its experimental shot against the best product on the market.
In the large trial Moderna had compared its new vaccine with Fluarix, an approved flu shot from the company GSK.
Moderna said the rejection was "inconsistent with previous written communications" with the FDA.
In a statement Wednesday Moderna's CEO, Stephane Bancel, said "we appreciate the FDA's engagement" in a "constructive" meeting the company had requested following the rejection.
"Pending FDA approval, we look forward to making our flu vaccine available later this year so that America's seniors have access to a new option to protect themselves against flu."
Since Trump retook the White House, both he and health chief Kennedy have come under broad criticism from public health and medical experts for sowing doubt about the safety and efficacy of vaccines widely known to be safe, and upending the pediatric immunization schedule.
Nogueira--PC
