-
 Kohli fires Bengaluru to big win after IPL remembers stampede dead
Kohli fires Bengaluru to big win after IPL remembers stampede dead
-
Graou shines as Toulouse sink Montpellier, Pau climb to second in Top 14

-
 Vingegaard nears Tour of Catalonia victory with stage six win
Vingegaard nears Tour of Catalonia victory with stage six win
-
Malinin bounces back from Olympic meltdown with third straight world skating gold

-
 French police foil Paris bomb attack outside US bank
French police foil Paris bomb attack outside US bank
-
Senegal parade AFCON trophy at Stade de France, despite being stripped of title

-
 Graou shines as Toulouse sink Montpellier to extend Top 14 lead
Graou shines as Toulouse sink Montpellier to extend Top 14 lead
-
Anti-Trump protests launch on 'No Kings' day in US

-
 Protesters rally in London against UK far-right rise
Protesters rally in London against UK far-right rise
-
France foils Paris bomb attack outside US bank

-
 Indian Premier League cricket season begins with silence to honour stampede dead
Indian Premier League cricket season begins with silence to honour stampede dead
-
Missing Cuba-bound aid boats located, crew reported safe

-
 Ignore our celebrations, we respect Bosnian team, says Italy's Dimarco
Ignore our celebrations, we respect Bosnian team, says Italy's Dimarco
-
Case closed for Morocco despite Senegal Afcon outrage

-
 22 migrants die off Greece after six days at sea: survivors
22 migrants die off Greece after six days at sea: survivors
-
Henderson backs England's White after Wembley boos

-
 Zelensky visits UAE, Qatar for air security talks with Gulf
Zelensky visits UAE, Qatar for air security talks with Gulf
-
Hollingsworth upsets Hunter Bell as Gout Gout fails to fire in Melbourne

-
 Iran footballers pay tribute to victims of school strike
Iran footballers pay tribute to victims of school strike
-
Questions over Israel's interceptor stockpiles as Mideast war drags on

-
 Sweet heist? Nestle says 12 tonnes of KitKat stolen
Sweet heist? Nestle says 12 tonnes of KitKat stolen
-
Pope denounces widening gap between the rich and poor on Monaco visit

-
 Yemen's Houthi enter war with missile targeting Israel
Yemen's Houthi enter war with missile targeting Israel
-
USS Gerald Ford arrives in Croatia for maintenance

-
 Antonelli leads Mercedes 1-2 as Verstappen suffers qualifying shock
Antonelli leads Mercedes 1-2 as Verstappen suffers qualifying shock
-
Verstappen calls his Red Bull 'undriveable' after more woes

-
 Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2
Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2
-
Millions angry with Trump expected to fill American streets

-
 Attacks across Middle East as Iran war enters second month
Attacks across Middle East as Iran war enters second month
-
Late surge lifts Thunder, Celtics rally to down Hawks

-
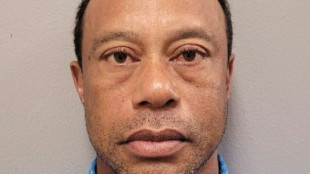 Tiger Woods arrested, charged with DUI after Florida crash
Tiger Woods arrested, charged with DUI after Florida crash
-
Antonelli leads Mercedes one-two in final Japan practice

-
 Unease for Iranian-Canadians after shooting at ayatollah critic's gym
Unease for Iranian-Canadians after shooting at ayatollah critic's gym
-
Sequins, slogans, conspiracies: Inside the right-wing culture at CPAC

-
 NBA fines T-Wolves center Reid $50,000 for ripping refs
NBA fines T-Wolves center Reid $50,000 for ripping refs
-
Sinner ousts Zverev to book Miami Open final with Lehecka

-
 McKellar hails 'special memory' after Waratahs stun Brumbies
McKellar hails 'special memory' after Waratahs stun Brumbies
-
Tuchel takes positives from scrappy England draw against Uruguay

-
 Japanese star Sakamoto signs off with fourth world skating gold
Japanese star Sakamoto signs off with fourth world skating gold
-
Tuchel disappointed after England fans boo White

-
 US envoy hopeful on Iran talks as strikes target nuclear facilities
US envoy hopeful on Iran talks as strikes target nuclear facilities
-
Controversial African champions Morocco salvage Ecuador draw on Ouahbi debut

-
 Dutch end Norway's unbeaten run as Haaland rests
Dutch end Norway's unbeaten run as Haaland rests
-
'Strait of Trump': US president says Iran must open key waterway

-
 Wirtz steals show as Germany win thriller in Switzerland
Wirtz steals show as Germany win thriller in Switzerland
-
White jeered on England return as Uruguay snatch friendly draw

-
 Tiger Woods arrested, charged with DUI after Florida crash: police
Tiger Woods arrested, charged with DUI after Florida crash: police
-
Oyarzabal double fires Spain to win over Serbia

-
 More to IOC gender testing than appeasing Trump: ex-IOC executive
More to IOC gender testing than appeasing Trump: ex-IOC executive
-
Japan's Sakamoto ends career with fourth world skating title


Northway Biotech Launches Full-Service Viral Clearance Studies, Delivering Results Faster Than Industry Standards
With six newly established, identical BSL-2 laboratories now operational, biologics CDMO Northway Biotech can conduct VCS programs for up to six clients simultaneously, significantly alleviating current market bottlenecks
VILNIUS, LT / ACCESS Newswire / May 5, 2025 / Northway Biotech, a biopharmaceutical contract development and manufacturing organization (CDMO), today announced the expansion of its protein-based and gene therapy service offerings with the addition of Viral Clearance Studies (VCS) capabilities. This strategic growth follows the opening of Northway Biotech's new Gene Therapy Center with dedicated cGMP facilities for virus-related projects.
With six newly established, identical BSL-2 laboratories now operational, Northway Biotech can conduct VCS programs for up to six clients simultaneously, significantly alleviating current market bottlenecks. Additionally, the company has expanded its capabilities to perform GMP-compliant manufacturing and testing under BSL-3 conditions, further strengthening its service offering across gene therapy and broader biologics development.
Viral Clearance Studies are now offered both as part of Northway Biotech's integrated CDMO programs and as a standalone service. This flexibility allows external clients to access VCS expertise independently, without requiring a manufacturing agreement.
Accelerated Delivery Timelines - Over One Month Faster Than Industry
Leveraging expanded infrastructure and integrated analytical capabilities, Northway Biotech is positioned to deliver Viral Clearance Studies substantially faster than the current industry standard. Comprehensive studies, assessing viral removal and inactivation, can now be completed with final regulatory-compliant reporting in under 10 weeks from initiation of project design when two model viruses are employed, and within 12 weeks when four model viruses are used.
"Our expansion into Viral Clearance Studies is a natural extension of our CDMO services, enabling us to manage these critical studies in-house and significantly reduce project timelines for our clients," said Prof. Vladas Algirdas Bumelis, CEO of Northway Biotech. "By investing in state-of-the-art BSL-2 and BSL-3 facilities, expanding technical capabilities, and further strengthening our scientific teams, we are uniquely positioned to deliver high-quality VCS data faster - a key advantage for clients advancing through clinical development and regulatory approval."
For more information on Northway Biotech's Viral Clearance Study processes, service offerings, and delivery timelines, please complete the contact form to connect with the Northway Biotech team.
About Northway Biotech - https://www.northwaybiotech.com
Northway Biotech is a leading contract development and manufacturing organization (CDMO) supporting customers worldwide. Its highly experienced and professional team executes projects at every stage, from cell line construction and process development to cGMP manufacturing of biopharmaceutical products. The company's extensive expertise and vertically integrated service offering enables rapid execution of multiple projects from its state-of-the-art GMP facilities while ensuring full process and product compliance at all stages of research, development, and commercial manufacturing. Northway Biotech is a privately owned company founded in 2004 and operates locations in Vilnius, Lithuania; London, United Kingdom; and Waltham, MA, USA.
Media & Business Contact:
Prof. Vladas Algirdas Bumelis
CEO and Chairman of the Board
Northway Biotech
[email protected]
Contact Information
Vladas Bumelis
CEO and Chairman of the Board
[email protected]
SOURCE: Northway Biotech
View the original press release on ACCESS Newswire
A.Silveira--PC




